What Is The Cost To Get Cast Iron Auto Parts Cleaned By Electrolysis
Cast Iron Cleaning With Electrolysis
Among the variety of restoration tools available to the dedicated vintage cast fe cookware collector, perhaps the well-nigh useful of all is cleaning by electrolysis. While a bit more work and expense than other methods to set up, a properly designed and implemented electrolysis tank can remove both rust and build-up in relatively short order.
The term electrolysis comes from 2 Greek words, and essentially means "to break down using electricity". Some may recall high school science grade experiments in which electrolysis was demonstrated to intermission downwards water into its molecular components of hydrogen and oxygen. But an electrolytic cell can also bear on the electrodes to which the voltage source is fastened, either past calculation cloth, removing material, or both. The process, under conditions of high voltage and temperature, is the ground for electroplating such equally that of decorative chrome on auto parts.
For our purposes, electrolysis cleaning works somewhat similar chrome plating in contrary. Past connecting the positive and negative wires the opposite of the plating process, you lot get grime and rust removal.
The most common set for an electrolysis iron cleaning tank involves a plastic storage container or the like, sturdy enough to be capable of belongings 8 or more gallons of water, and a car bombardment charger. You'll need a piece of metal, either atomic number 26 or steel, that will serve equally a "sacrificial anode" to which the electric current will flow from the piece being cleaned.
You lot'll as well demand turn the h2o in the tank into what's chosen an electrolyte, making information technology more conductive so the electric current volition flow more readily through it. For this, we apply Arm & Hammer Super Washing Soda™ (not baking soda), available in the laundry additives department (medium size yellowish box), at the rate of 1-2 tablespoons per gallon of water. Washing soda is primarily sodium carbonate, whereas baking soda is sodium bicarbonate. Some people employ a swimming puddle h2o conditioner called pH+, which consists of sodium carbonate. Some advanced hobbyists use sodium hydroxide aka lye to attain a double-acting electrolyte/cleaning solution, just for nearly, the simpler and less-chancy washing soda volition do.
| Gallons of Water | Washing Soda | |
|---|---|---|
| five | 5-10 T. | ane/3 - 2/3 C. |
| x | ten-twenty T. | two/3 - 1-one/3 C. |
| xv | 15-30 T. | one - ii C. |
| twenty | 20-forty T. | 1-ane/3 - 2-ii/three C. |
| 25 | 25-fifty T. | 1-2/iii - 3-1/3 C. |
To properly connect the voltage source, you just have to remember the blacK (negative) wire goes on the sYardillet. Also, the battery charger you employ must be a manual one, or have a manual charge mode. An automatic charger volition see the electrolysis tank as a charged bombardment and shut itself down.
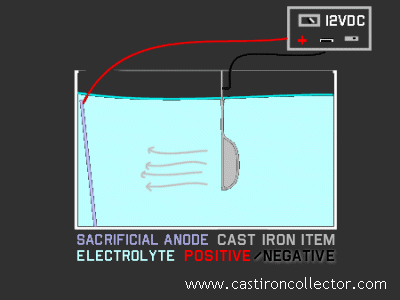
If you already own a fully automated charger and don't wish to purchase a manual one, there is a workaround, although it necessitates the apply of a 12V auto battery. Hooking up an automatic charger to the battery as if to charge it, you tin then use jumper cables from the battery to your electrolysis setup. Electric current stored in the battery volition flow to the pan and sacrificial metal, and the charger will happily supply current to the depleted bombardment. Heightened intendance is required using this setup as you must be diligent in properly maintaining the positive-to-positive and negative-to-negative connections between charger and battery. You must besides ensure that the positive and negative leads from the battery practice non straight bear upon. Additionally, the terminals and clamps tin become hot.
I use a 2amp/10amp switchable transmission  Die Hard™ Charger from Sears. I understand Sam's Gild has some inexpensive transmission chargers as well. I place a length of 2x2 wood across the tiptop of my container, and append the pans in the water from information technology with coat hanger wire, attaching the black connector to the unsubmerged tip of the handle of the pan. The other, cherry connector goes to a slice of air conditioner cabinet steel canvas metal I become from an HVAC guy who oft has panels of new, unpainted metal left over from his installations.
Die Hard™ Charger from Sears. I understand Sam's Gild has some inexpensive transmission chargers as well. I place a length of 2x2 wood across the tiptop of my container, and append the pans in the water from information technology with coat hanger wire, attaching the black connector to the unsubmerged tip of the handle of the pan. The other, cherry connector goes to a slice of air conditioner cabinet steel canvas metal I become from an HVAC guy who oft has panels of new, unpainted metal left over from his installations.
Other options for inexpensive anodes include rebar or used lawn mower blades. Larger steel cans like those for fruit juices with pinnacle and bottom removed, cut down the side and flattened are another cheap alternative. Anodes with more surface area tend to be the most efficient.
For best results, make certain the connectors make adept electric contact with both the piece being cleaned and the sacrificial metallic. Utilise your wire brush or stainless steel scrubber to remove some of the rust and/or crud at the spot on your piece to which you will be attaching the charger connector. In the long term, to go along your clamps away from corrosive moisture or being affected past the electrolytic process, you may opt to non connect your charger clamps directly to the piece, instead attaching them to the metal subclass or wire the piece is hung from. Adequate current should however exist able to menstruum if all zipper points are relatively clean, bare metallic. Poor connections crusade increased electrical resistance and excessive heat. Clean metal-to-metal connections will assure the most efficient cleaning and the least damage to charger leads over time. Charger clamps becoming noticeably hot during employ is an indication of poor contact.
Also, don't be tempted to add more washing soda than recommended; it tin cause excessive current and overheating bug which may brand the charger close downwardly or melt cable wire insulation. You'll know you lot take skilful current flowing when you lot meet a mist of fine bubbling forming effectually the piece and your charger's amp meter reads towards the upper end of its scale.
The process of electrolysis converts red rust (ferric oxide) to ferrous oxide, sometimes called black rust. The procedure also both coats and rots the "sacrificial" piece of metal over time, so it needs to exist scraped downwards occasionally, or flipped so that a clean side faces the piece being cleaned, and, eventually, replaced.
A byproduct of the electrolytic process is the formation of potentially flammable hydrogen gas. It is prudent, therefore, to insure the area effectually the setup is well-ventilated, or, meliorate, to consider doing information technology outdoors.
Electrolysis is largely a line-of-sight process, meaning the side of the slice closest to the sacrificial metal volition become cleaner first. If yous put something between the piece and the metal, a "shadow" of crud will be left on the piece where the object blocked the menses of current from the slice. Some people'south set-ups have metal on both sides, or surrounding the piece for faster action. I simply turn the piece around from fourth dimension to fourth dimension. Visually, built-upwardly crud loosens, and peels or flakes off like old paint. In some places, it sticks tighter, and takes longer to come off. The red rust will plough into a fine black residue that easily wipes or scrubs off. The process is finished when the metal is bare and grayness. Some darker staining may remain in spots that were especially cruddy, but that's OK, it tin be dealt with.
Tip: If the rust is only on the inside, large format pieces like cauldrons and washing kettles can become their own electrolysis tank. Fill with water and deliquesce an advisable corporeality of washing soda for the book. Use a 2x4, a piece of PVC piping or other non-conductive fabric as a batten and hang a piece of sacrificial metal from it. Attach the negative cablevision of a manual bombardment charger to the side of the pot and the positive to the sacrificial anode.
How long does the electrolysis take? Before I started using lye, cleaning an average piece using electrolysis alone might accept taken a couple of sessions, maybe eight hours each. Softening things up with the lye first reduces that to about one afternoon session of a few hours. Hanging the slice to be cleaned as shut as possible without touching the sacrificial metal too tends to speed upwards the process.




Ii identically rusted Lodge #7s, earlier and after electrolysis:


Other Thoughts
Equally y'all read about the usage of electrolysis for cast fe cleaning, yous will oftentimes encounter a few caveats almost the choice of materials for the sacrificial anode.
Many frequent electrolysis users, unhappy with the constant need for anode replacement, have turned to stainless steel, some fifty-fifty going so far as to create a 360° setup by using a stainless steel barrel as both container and anode. The advantage to stainless steel is that it does not corrode as readily as exercise other types of steel or fe. It is not unusual to run across comments, however, that using stainless steel in an electrolysis setup creates a chancy byproduct chosen hexavalent chromium. "Hex chrome", every bit it is referred to in the electroplating manufacture, is indeed a problem for those working in that industry, where, at the temperatures and voltages employed, it tin exist produced, vaporized, and released into the atmosphere. At the far lower voltages and temperatures unremarkably used for cast iron cleaning, notwithstanding, hex chrome is not a business.
Like warnings are seen against using galvanized metals, and the possibility that zinc may be released into the electrolyte, where information technology may come up into contact with the piece being cleaned. Again, the voltages used should not be sufficient to cause concern.
Proper disposal of used electrolyte, however, should include avoidance of soil contamination near vegetable gardens. And, as with any cleaning process, proper protocols should include thoroughly washing and rinsing the cleaned slice earlier beginning any seasoning regimen.
To avert the foregoing types of concerns birthday, the employ of graphite every bit an anode appears to fit the beak quite nicely. Graphite is a form of carbon that happens to be electrically conductive, but at the aforementioned fourth dimension is far less reactive to the electrolytic process than the majority of metals. Every bit such, the only thing that it tin possibly introduce back into the electrolyte or to the slice existence cleaned is manifestly carbon. Graphite as well has the advantage of not condign coated with iron oxide as practise typical metal anodes. Information technology therefore needs no regular cleaning to maintain its performance. Storing the anode dry between cleaning sessions is recommended.
Although non as inexpensive as plain sheet metallic or scrap iron, graphite can be had, considering its expected duty life, quite reasonably. Bars, rods, or plates of compressed extruded graphite are available from a multifariousness of sources. Await online for liquidation sales of lots of remaindered graphite forms, fugitive those which mention other materials like copper in their composition.
It is important to notation that, over time, any anode material used for electrolysis cleaning will deteriorate and will somewhen need to be replaced.
Finally, electrolysis should only be used to clean bare cast iron ware. Aluminum pieces will dissolve. Enameled cast iron pieces may also have their coatings compromised. Chrome or nickel-plated iron pieces may or may not be adversely affected, depending upon whether or non areas of plating have already begun to release or chip off.
Source: https://www.castironcollector.com/electrolysis.php
Posted by: evanssiond1998.blogspot.com


0 Response to "What Is The Cost To Get Cast Iron Auto Parts Cleaned By Electrolysis"
Post a Comment